resDNASEQ CHO Residual DNA Quantitation kit
$1,899.00
Buy Our Ultra-trace CHO residual DNA quantitation kit with industry-leading 0.01 fg/μL LoD. 20-minute sample preparation, 1.5-hour total test time, prep components can be storaged with room-temperature. Fully aligned with WHO guidelines (10 ng per therapeutic dose), offering 70%-130% recovery rate and <30% CV. Ideal for biopharmaceuticals, monoclonal antibodies, and recombinant protein production QC. Get Free Trial & Discount Quote Today!
| Catalog number: | D24011202 |
|---|---|
| Qty: | 100Reactions |
| Include | Assay Kit only |
Description
CHO Residual DNA Quantitation kit
The Ducky Bio CHO Residual DNA Quantitation Kit enables precise, reliable quantification of residual host cell DNA (resDNA) from Chinese Hamster Ovary (CHO) cell lines, which are pivotal in biopharmaceutical manufacturing.
Based on robust real-time quantitative PCR (qPCR) technology, this assay delivers rapid, specific, and highly sensitive detection of CHO-derived DNA. It is meticulously designed to target unique genomic sequences of the CHO cell line, ensuring exceptional specificity. This design effectively eliminates the risk of false positives from non-target DNA, such as human or environmental contaminants that may be introduced during sample handling and processing.
Developed to meet and exceed rigorous regulatory standards, the kit’s sensitivity is calibrated to comply with the WHO guideline of ≤10 ng residual DNA per therapeutic dose, providing confidence for your product safety and quality control protocols.
Unmatched Trace-Level Sensitivity
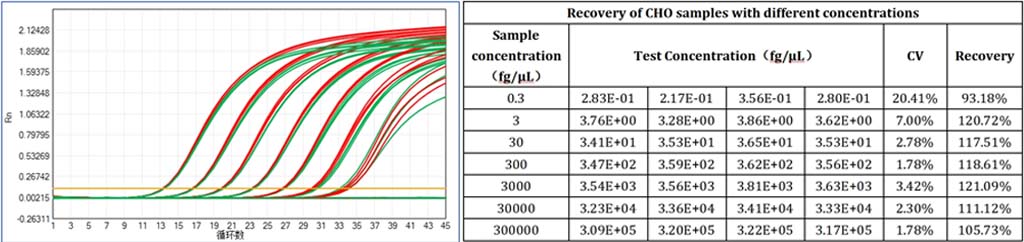
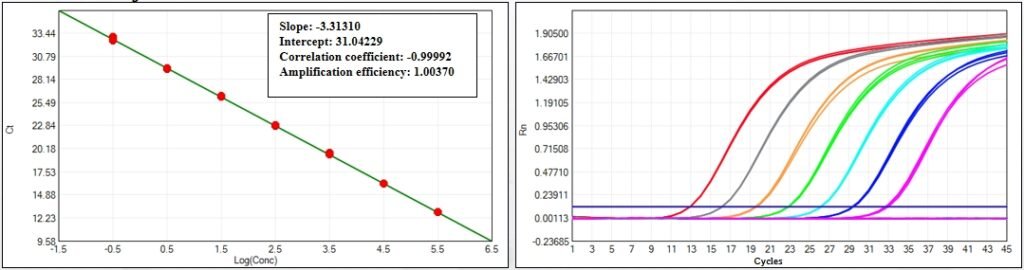
Setting a new benchmark for CHO residual DNA detection, this kit achieves a LoD of 0.01 fg/μL and LoQ of 0.3 fg/μL—far exceeding industry averages and ensuring even trace contaminants are captured. Backed by a regression coefficient of 0.99992 and amplification efficiency of 100.370%, it delivers consistent results across the entire linear range (0.3 fg/μL to 300 pg/μL), with CV values <30% for all concentrations ≥0.3 fg/μL. For biomanufacturers targeting ultra-pure products, this level of sensitivity eliminates the risk of regulatory non-compliance.
Workflow Revolution for QC Efficiency
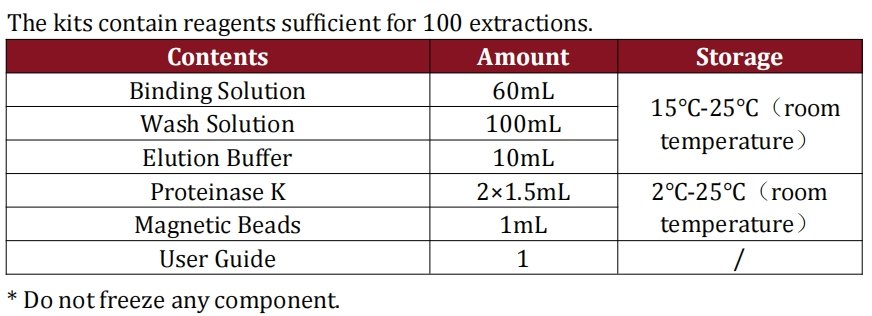
We’ve reimagined sample preparation to cut down on time and logistical hassle. With just 3 intuitive steps (Binding → Washing → Elution) completed in 20 minutes, the process eliminates complex protocols and specialized training. What’s more, every component of the sample preparation suite—from Binding Solution to Magnetic Beads—stays stable at room temperature (15°C-25°C), removing the need for expensive cold chain storage and simplifying inventory management. Paired with a single-tube qPCR MIX, the entire assay (from sample prep to results) wraps up in 1.5 hours—slashing traditional QC timelines by up to 60%.

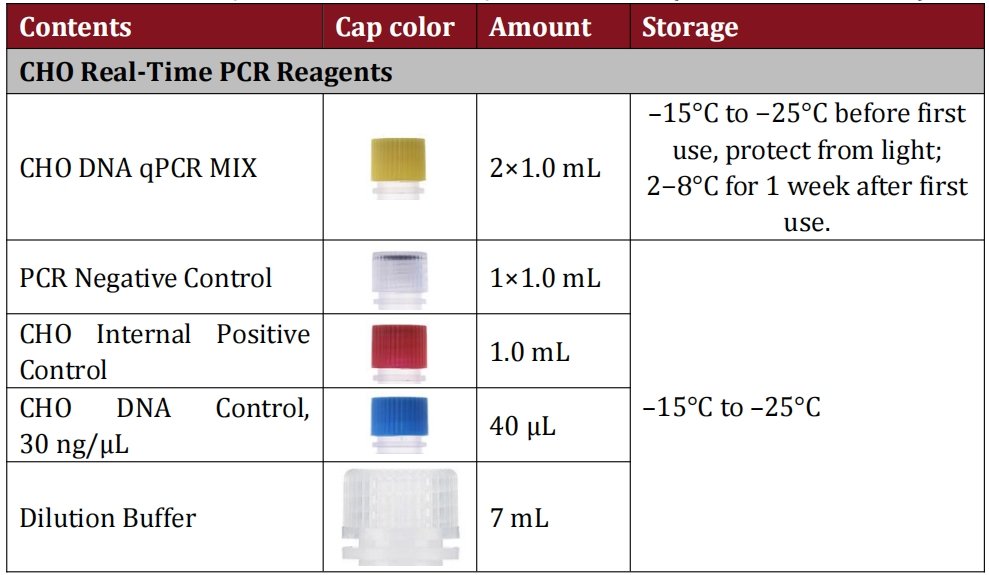
Regulatory Confidence & Absolute Specificity
Compliance with WHO, FDA, EMA, and ICH guidelines is baked into every aspect of this kit. It’s rigorously validated to meet the 10 ng CHO DNA per therapeutic dose threshold, giving you peace of mind during regulatory inspections. Unlike competing kits that may cross-react with human or environmental DNA, our assay targets unique CHO cell DNA sequences—ensuring that every result accurately reflects only residual CHO DNA levels, even in complex biotherapeutic matrices.
Robust Performance Across Diverse Samples
Whether you’re testing cell culture supernatants, bulk drug substances, or final biotherapeutic formulations, this kit delivers reliable quantification. Spiked sample validation confirms a recovery rate between 70% and 130%, demonstrating its ability to perform consistently in real-world sample conditions. The kit’s 100.370% amplification efficiency ensures sharp, reproducible curves—even for ultra-low concentration samples—making it a trusted tool for both process development and routine QC.
What Makes Our CHO Residual DNA Kit Stand Out?
At Ducky Bio, we didn’t just build a kit—we engineered a solution tailored to the unique challenges of CHO cell-derived bioproduction. Unlike one-size-fits-all residual DNA assays, our resDNASEQ kit is optimized exclusively for CHO DNA, leveraging proprietary primer-probe designs to eliminate cross-reactivity. The room-temperature stable sample prep components reduce logistical costs and waste, while the 1.5-hour total assay time frees up lab resources for high-priority tasks.
We also prioritize transparency: every kit undergoes rigorous batch-to-batch testing to ensure consistent performance, with detailed validation data included to support your regulatory submissions. For biopharmaceutical teams seeking a balance of sensitivity, speed, and compliance, this kit isn’t just a tool—it’s a strategic advantage in a highly regulated industry.
Applications
>Monoclonal antibody (mAb) production process QC
>Recombinant protein and fusion protein manufacturing
>CHO cell line-based vaccine development and production
>Biotherapeutic bulk drug substance and final product testing
>Cell culture process optimization and scale-up validation
>Regulatory compliance testing for global biopharmaceutical markets
>Impurity control in CHO-derived biotherapeutics
You May Also Interested
Frequently Asked Questions (FAQs)
1.What sets this CHO residual DNA kit apart from other qPCR-based assays?
Answer: Our kit offers an industry-leading LoD of 0.01 fg/μL—far more sensitive than many competing products—and features absolute specificity for CHO cell DNA (no cross-reactivity with human or environmental DNA). Additionally, all sample prep components are room-temperature stable, eliminating cold storage costs, and the 20-minute sample prep + 1.5-hour total assay time delivers unmatched workflow efficiency.
2.Can this kit be used for both process development and routine QC?
Answer: Yes—its broad linear range (0.3 fg/μL to 300 pg/μL) and consistent performance make it ideal for both process optimization (tracking residual DNA during scale-up) and routine QC testing of final products. The kit’s compliance with WHO and GMP standards also supports regulatory submissions for biotherapeutics.
3.Do I need specialized equipment or training to use this kit?
Answer: No— the kit uses standard real-time qPCR instruments (compatible with most commercial platforms) and requires only basic lab skills. The 3-step sample prep is intuitive, and the single-tube qPCR MIX reduces hands-on time and human error. We also provide a detailed user guide, and our technical support team is available to assist with any questions.
4.How stable are the kit components during shipping and storage?
Answer: Sample prep components are designed for room-temperature storage and shipping—no dry ice required, which reduces logistics costs. The qPCR reagents are shipped on dry ice and remain stable at -15°C to -25°C for the kit’s shelf life (see lot-specific expiration date). After first use, the qPCR MIX can be stored at 2-8°C for up to 1 week without losing performance.
5.Is this kit suitable for testing complex biotherapeutic formulations (e.g., antibody-drug conjugates)?
Answer: Absolutely. The kit’s robust recovery rate (70%-130%) and resistance to matrix interference make it effective for testing a wide range of biotherapeutic samples, including antibody-drug conjugates, fusion proteins, and concentrated protein formulations. We recommend following the sample preparation protocol in the user guide to ensure optimal results.
For more details on technical validation, customization options, or to request a personalized quote, contact our team today. Ducky Bio is dedicated to delivering innovative, reliable residual DNA testing solutions that empower biopharmaceutical manufacturers to meet regulatory standards and accelerate product development.
Related products
-
Mycoplasma Detection Kit(qPCR)
$1,799.00 -
Pichia Pastoris resDNA Assay Kit
$1,899.00 -
Sample Preparation Kit Ⅱ
$899.00